Development of lipid bilayer platforms
One of our major focuses is the development and application of artificial cell membrane platforms, also known as lipid bilayer platforms. These technologies have received much interest as models of cell membranes, allowing scientific study of the properties of cell membranes, membrane proteins, and the interaction of the membrane and proteins to external chemical stimuli (such as interaction to pharmaceutical compounds). They have also sparked much interest as chemical sensors, with applications from novel techniques for DNA sequencing to extremely fast, highly sensitive, single molecule measurement of ion transport across cell membranes. The short lifetime and fragility of the biological membranes into which channel proteins must be incorporated have proven to be fundamental challenges to the practical development of these technologies. To confront these challenges, the group investigates novel approaches to membrane synthesis and stabilization, including innovative techniques for rapid and reliable parallel membrane formation, membrane mechanical stabilization by hydrogel support, and automated membrane formation in microfluidic devices. These complementary membrane engineering strategies are highly innovative and represent significant advances in the state of the art of membrane technology.
One of our major focuses is the development and application of artificial cell membrane platforms, also known as lipid bilayer platforms. These technologies have received much interest as models of cell membranes, allowing scientific study of the properties of cell membranes, membrane proteins, and the interaction of the membrane and proteins to external chemical stimuli (such as interaction to pharmaceutical compounds). They have also sparked much interest as chemical sensors, with applications from novel techniques for DNA sequencing to extremely fast, highly sensitive, single molecule measurement of ion transport across cell membranes. The short lifetime and fragility of the biological membranes into which channel proteins must be incorporated have proven to be fundamental challenges to the practical development of these technologies. To confront these challenges, the group investigates novel approaches to membrane synthesis and stabilization, including innovative techniques for rapid and reliable parallel membrane formation, membrane mechanical stabilization by hydrogel support, and automated membrane formation in microfluidic devices. These complementary membrane engineering strategies are highly innovative and represent significant advances in the state of the art of membrane technology.
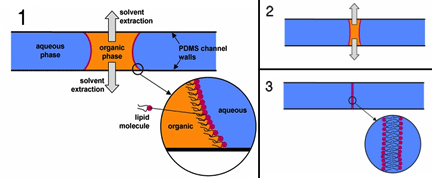
However, these applications are often hindered by the short lifetime and fragility characteristic of freestanding artificial lipid bilayer membranes. Our group aims to address these shortcomings through the development of improved bilayer synthesis, rapid and reliable parallel membrane formation and measurement, hydrogel stabilization, and integration with automation and microfluidic technologies.
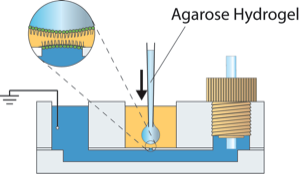
Improved lipid bilayer technologies may lead to their wider application. Increased measurement throughput resulting from parallelism, automation, or high-speed solution exchange. To explore this potential, we have integrated lipid bilayer arrays with a Tecella multi-channel patch clamp amplifier to simultaneously measure up to 48 bilayers in parallel. We have also constructed apparatus integrating a hydrogel-supported bilayers with a fluidic pump to allow the simultaneous measurement during rapid flow of the surrounding solution.
References
- “Hydrogel-Stabilized Droplet Bilayers for High Speed Solution Exchange,” Shiv A. Acharya, Alexander Portman, Carl S. Salazar, Jacob J. Schmidt, Scientific Reports 3 : 3139, DOI: 10.1038/srep03139 (2013)
- “Microfluidic Passive Permeability Assay using Nanoliter Droplet Interface Lipid Bilayers,” Takasi Nisisako, Shiva Portonovo, Jacob Schmidt, Analyst 138(22), 6793-6800 (2013)
- “hERG Drug Response Measured in Droplet Bilayers,” Shiva A. Portonovo, Carl S. Salazar, and Jacob J. Schmidt, Biomedical Microdevices 2013, 15 (2), 255-259 (2012)
- “Ion channel drug potency assay with an artificial bilayer chip,” Ahmad M. El-Arabi, Carl J Salazar, and Jacob Schmidt, Lab Chip 2012, 12, 2409-2413
- “Masking apertures enabling automation and solution exchange in sessile droplet lipid bilayers,” Shiva A. Portonovo and Jacob Schmidt, Biomed. Microdev. 14, 187-191 (2012)
- “Automatable lipid bilayer formation and ion channel measurement using sessile droplets,” J L Poulos, S A Portonovo, H Bang, J J Schmidt, J. Phys.: Condens. Matter 22 (2010) 454105
- “Automated lipid bilayer and ion channel measurement platform,” Tanuj Thapliyal, Jason L. Poulos, and Jacob J. Schmidt, Biosensors and Bioelectronics 26 (2011), 2651-2654z, doi:10.1016/j.bios.2010.01.017 (2010)
- “Automatable production of shippable bilayer chips by pin tool deposition for an ion channel measurement platform,” Jason L. Poulos, Tae-Joon Jeon, Jacob J. Schmidt, Biotechnology Journal 5 (5), 511-514 (2010).
- “Electrowetting on dielectric-based microfluidics for integrated lipid bilayer formation and measurement,” Jason L. Poulos, Wyatt C. Nelson, Tae-Joon Jeon, Chang-Jin "CJ" Kim, and Jacob J. Schmidt, Applied Physics Letters 95, 013706 (2009)
- “Ion Channel and Toxin Measurement Using a High Throughput Lipid Membrane Platform,” Jason L. Poulos, Tae-Joon Jeon, Robert Damoiseaux, Eugene J. Gillespie, Kenneth A. Bradley, Jacob J. Schmidt, Biosensors and Bioelectronics 24, 1806-1810 (2009) doi:10.1016/j.bios.2008.08.041
- “Long-term storable and shippable lipid bilayer membrane platform,” Jason L. Poulos, Tae-Joon Jeon, Robert Damoiseaux, Tae-Joon Jeon, Jason L. Poulos and Jacob J. Schmidt, Lab on a Chip, 8, 1742-1744 (2008)
- “Black lipid membranes stabilized through substrate conjugation to a hydrogel,” Tae-Joon Jeon, Noah Malmstadt, Jason L. Poulos, Jacob J. Schmidt, Biointerphases 3(2), FA96-FA100 (2008)
- “Long-Lived Planar Lipid Bilayer Membranes Anchored to an In Situ Polymerized Hydrogel,” N. Malmstadt, T.-J. Jeon, J. J. Schmidt, Advanced Materials 20 (1), 84-89 (2008)
- “Automated Formation of Lipid-Bilayer Membranes in a Microfluidic Device,” Noah Malmstadt, Michael A. Nash, Robert F. Purnell, and Jacob J. Schmidt, Nano Lett. 6(9), 1961-1965 (2006)
- “Single molecule measurements of channel proteins incorporated into biomimetic polymer membranes,” Denise Wong, Tae-Joon Jeon and Jacob Schmidt, Nanotechnology 17 3710-3717 (2006)
- “Hydrogel-Encapsulated Lipid Membranes,” Tae-Joon Jeon, Noah Malmstadt, and Jacob J. Schmidt, Journal of the American Chemical Society 128, 42-43 (2006).